News Story
Mapping Nucleation Kinetics with Nanometer Resolution
Nucleation is the formation of a new condensed phase from a fluid phase via self-assembly. This process is critical to many natural systems and technical applications including the manufacturing of pharmaceuticals and advanced materials, the formation of clouds, mineral formation in the Earth’s crust, and the stability of proteins. While scientists have studied nucleation for over a century, it remains an elusive process because it occurs sporadically in time. Moreover, a nucleus can be less than a nanometer (10-9 m) in size.
Thus far, indirect methods and simulations have been used to study nucleation – literally watching the nucleation process occur has been beyond scientific reach. One challenge is that the process almost always occurs at a solid-liquid interface, making nucleation more energetically favorable. However, the identity of the nucleation sites (i.e., their shape and chemistry) is almost never known - meaning the rules dictating where the process will occur, and how fast, are unknown - making it difficult to predict the outcome, thus, limiting scientists' ability to accurately predict mineral transformations in the Earth’s crust, weather patterns, and optimal conditions for synthesis of advanced materials.
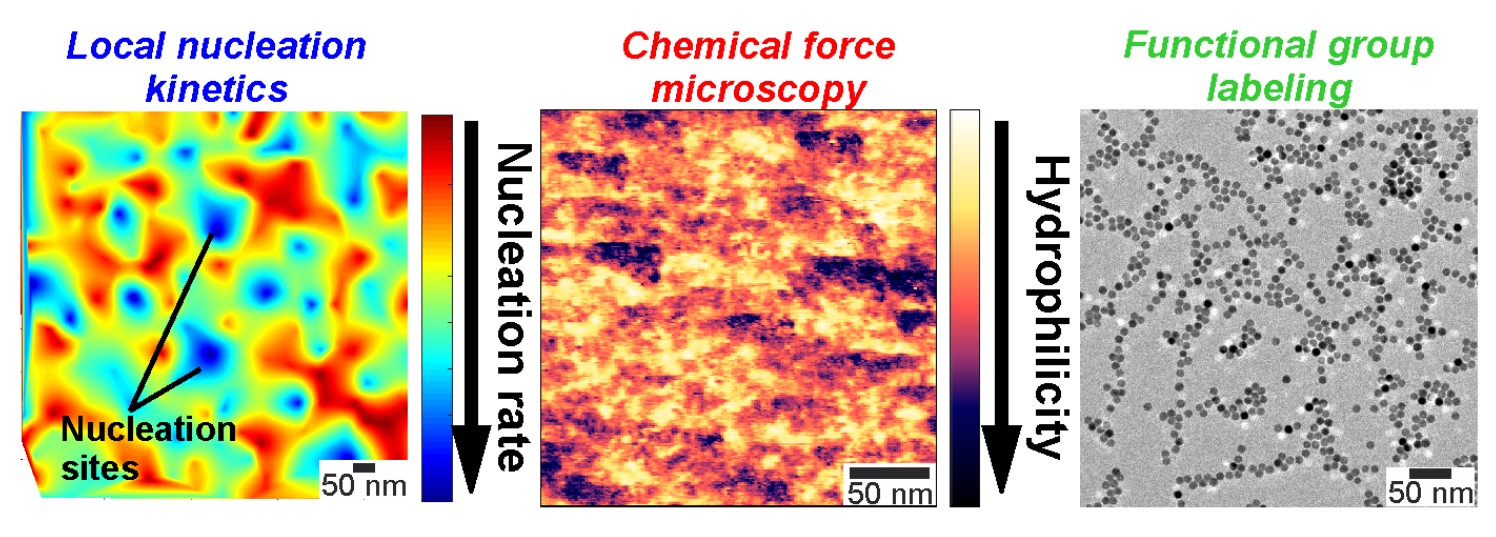
To that end, researchers in the University of Maryland (UMD) Department of Chemical and Biomolecular Engineering (ChBE) have recently published a study in the Journal of the American Chemical Society detailing the discovery of a new imaging method – nanoscale nucleation kinetic mapping – which allows direct imaging of nucleation events at a solid-liquid interface. Through the use of scanning transmission electron microscopy and an environmental microfluidic cell (LC-STEM), the group – led by ChBE Assistant Professor Taylor Woehl – witnessed heterogeneous nucleation at a silicon nitride-water interface. Mei Wang, a ChBE Ph.D. Student, served as first author of the study. This study represents one of the first cases where the kinetics of nucleation have been directly connected to the identity of the nucleation sites.
“Through the creation of nanoscale maps showing the local nucleation kinetics – in other words, how fast nuclei form locally – we found that nucleation occurred preferentially at discrete, nanometer sized regions of the interface,” said Dr. Woehl. “The most intriguing aspect was how non-uniform the nucleation kinetics were on the macroscopically flat, homogeneous interface. An in-depth study of the interface revealed the distribution of surface chemical groups to be highly non-uniform. Together with a theoretical model, our results showed that heterogeneous nucleation occurred preferentially on these domains of surface chemical groups.”
Broadly, this research has two outcomes: first, it reveals that nucleation kinetics can be non-uniform at a solid-liquid interface that appears to be uniform on the macro scale.
Secondly, the study introduces a new electron microscopy technique capable of following heterogeneous nucleation at a solid-liquid interface with nanometer scale resolution.
“We expect our study will have important implications in several research fields,” Woehl said. “This research pushes the boundaries of our current understanding of nucleation at complex solid-liquid interfaces – such interfaces include those on aerosol particles that cause cloud formation through nucleation of water droplets, or mineral-water interfaces in the Earth’s crust where deposition of new minerals occurs via heterogeneous nucleation.”
Wang adds that “Our results showed that variations in native surface chemistry on a uniform interface can significantly affect nucleation kinetics of nanocrystals. This finding is not only important in the transmission electron microscopy community for studying the formation kinetics of nanocrystals, but can also provide new insights for other technological processes involving surface crystallization, such as synthesis of nanomaterials or advanced energy materials like halide perovskite solar cells and battery electrodes.”
For additional information:
Wang, M. et al (2019, 30 July). “Nanoscale Mapping of Nonuniform Heterogeneous Nucleation Kinetics Mediated by Surface Chemistry,” Journal of the American Chemical Society. DOI: 10.1021/jacs.9b05225
Published August 20, 2019